/GettyImages-769723031-5a75818beb97de0037db7c78.jpg)
So we see monoxide or pentoxide, rather than monooxide or pentaoxide in molecule names. In some cases, when a prefix ends in a or o and the element name begins with o, we drop the a or o on the prefix. A Roman numeral in parentheses, preceded by the name of the element, is used for elements that can form more than one positive ion. The nomenclature, or naming, of ionic compounds is based on the names of the component ions. Different compounds have different names. Ionic compounds consist of cations (positive ions) and anions (negative ions). They are named following a different rule from ionic compounds. Because more than one atom of each element is present, prefixes are. Its name, if you go through all the steps, is sulfur trioxide. Chemical Nomenclature for Non-metal Compounds '(prefix, not mono)less anion-like atom name + (prefix)more anion-like atom name-ide' Non-metal compounds are often called covalent compounds. Because nitrogen is to the left of oxygen in the periodic table, nitrogen is named first. The first uses prefixes to indicate the number of atoms of an element that. Rule A-1.1 of the IUPAC Organic Nomenclature. Naming molecules in chemistry There are two systems of naming molecular compounds. The system of chemical nomenclature is designed to give this compound its own unique name. Numerical terms are used in chemical names for indicating a number of identical structural units in a compound. 
SO 3 has three oxygen atoms in it, so it is a different compound with different chemical and physical properties. Why all this trouble? There is another common compound consisting of sulfur and oxygen whose molecular formula is SO 3, so the compounds need to be distinguished. 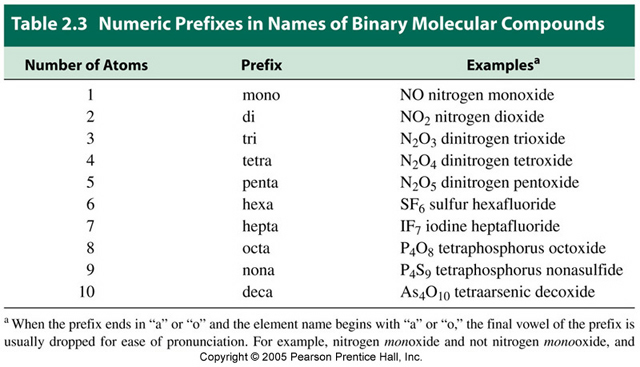
Bringing these two words together, we have the unique name for this compound-sulfur dioxide. Trivial names are well known for some molecular compounds and they are often used in the place of systematic names, e.g., water for H 2O and ammonia for NH 3.\)) with the stem ox- and the suffix -ide, to make dioxide. Examples of the names are: NO is nitrogen monoxide, CO 2 is carbon dioxide, PCl 3 is phosphorous trichloride, P 2O 5 is diphosphorus pentoxide, SiO 2 is silicon dioxide. Table 3.5.1 lists the prefixes used to represent the number of atoms from 1 to 10. Traditionally the suffix -ium was used only for metals (or at least elements that were expected to be metallic), and other elements used different suffixes. Several examples are found in Table 3.3.1. The atoms of a polyatomic ion are tightly bonded together and so the entire ion behaves as a single unit. If applicable, add a prefix (mono-, di-, tri-, etc.) to indicate the number of atoms of the element in the compound. The formula of the carbonate ion is CO 32. Use the element name in the periodic table. HOWEVER the prefix mono- is never used to name the first element To. The carbonate ion (see figure below) consists of one carbon atom and three oxygen atoms and carries an overall charge of 2. Examples of monatomic ions include Na +, Fe 3 +, Cl, and many, many others. This differentiates polyatomic ions from monatomic ions, which contain only one atom. The prefix poly- means many, so a polyatomic ion is an ion that contains more than one atom. If the prefix ends with a vowel and the element name begins with a vowel, drop the ending vowel of the prefix. element furthest left in the periodic table) and then the electronegative species. In this article, we will discuss polyatomic ions.Do not write mono- if it applies to the first element in the formula, but write mono- if it applies to the second element. Appendices: Periodic Table Units Constants Equations Reduction Potentials Elements and their Properties.the name of the second element with a prefix showing the number of atoms and its last syllable replaced with –ide.name of the first element in the formula with a prefix showing the number of atoms, followed by,.The name of binary covalent compounds contains prefixes, listed in Table 1, to indicate the number of atoms followed by the name of the elements according to the following rules: The first word is the name of the first element. Writing the names of binary covalent compounds When you have two different elements, there are usually only two words in the compound name.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
